Media o nas
ERA test
What is the endometrium?
The interior of the uterus is lined with a tissue called endometrium, which is prepared each month for the arrival of an embryo and is the place where the embryo implant and resides during gestation. When this does not occur, menstruation begins, shedding the endometrial lining.
Problems with endometrial lining are one of the leading causes of infertility in women.
Endometrial Receptivity
Sometimes embryo implantation in the endometrium does not develop successfully. The failure can occur for different reasons. A particularly important factor is endometrial receptivity. Endometrium is receptive when is ready for embryo implantation to occur, which usually takes place between days 19-21 of the menstrual cycle (5-7 days post-ovulation). This period of receptivity is called the window of implantation.
Evaluation of endometrial receptivity is particularly important to diagnose causes of infertility and miscarriages.
What is ERA?
The Endometrial Receptivity Array (ERA) is a novel diagnostic method developed and patented by IGENOMIX R&D, pioneers in reproductive genetics.
ERA allows to evaluate the endometrial receptivity status of a woman, from a molecular point of view. The biopsy of endometrial tissue is performed by a gynaecologist easily and quickly. An ERA predictor analyzes the collected data and classifies the endometrium as 'receptive' or 'non-receptive'. The results of the analysis determine whether or not the patient will be responsive to embryo implantation at the time of sampling. In case that the stage in non-receptive, the test allows to find a personalized window of implantation for each patient. Thus, ERA increases chances of successful in Vitro.
Who should use the ERA test?
The ERA test has been proven in patients who have experienced implantation failures with good quality embryos (at least 3 implantation failures in yound women or 2 in patients 37 years-old or more). This test is also indicated for patients with an apparently normal uterus who have been trying to get pregnant with no success.
The ERA test is recommended and performed by the gynaecologist who inform the patient about the test result.
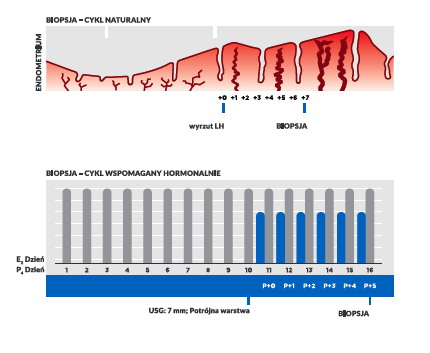 Procedure description
Procedure description
To carry out this analysis, a biopsy of the endometrium needs to be done at approximately day 21 of menstrual cycle. It is performed by the gynaecologist using thin cannula. There is no less invasive technique available to obtain a sufficient amount of endometrial material, thus some discomfort may be experienced, and slight bleeding may be noticed after the biopsy. However, this is the usual process with no added risks.
There is a risk (<5%) that the biopsy procedure fails to obtain a sufficient quantity and/or quality of endometrial tissue. In this case, a new biopsy will be required.

 00 48 600 466 592
00 48 600 466 592




